Alkalinity In Pools Whys It So Important And How To Control
Alkalinity in pools is an extremely important topic as its what helps prevent sudden pH changes. Total alkalinity is a measure of alkaline substances in this case, ionic compounds, that act as buffers that resist pH change.
Dont worry, this is just a bunch of chemistry and scientific mumbo jumbo that you really dont need to know. What is important to understand is that alkalinity helps stabilize the pH balance in your pool, and keeping it within normal range can save you a lot of headaches as well as solve a lot of problems.
The recommended range for a swimming pools total alkalinity is between 80 and 120 parts per million. Any good pool testing kit will let you determine the range, and its important to test often.
So, when it comes to alkalinity there are really only 3 states in can be in: Too High, Too Low and Just Right.
Low Alkalinity With low alkalinity your pH levels can swing back and forth between too high, and too low. Its not uncommon for these numbers to shift drastically causing an unbalance in your pools pH.
Some problems that are associated with this include: -Chlorine inefficiency -Metal Corrosion -Pool Staining -Scale
You can easily raise a pools alkalinity by using a chemical called sodium bicarbonate which is, baking soda. The recommended dosage is 1.5 pounds of sodium bicarbonate per 10,000 gallons of water. If youre unsure how much water you have you can find a good estimate by using this formula:
Poolalkalinity Vs Ph Level
Figuring out how to raise alkalinity inpool water can sometimes be tricky. One of the most common misconceptions ishow we use pH level and alkalinity interchangeably. While they are bothnecessary in testing and keeping water chemistry, pH level and alkalinity arenot the same.
Water pH gauges hydrogen in the water tosee how acidic or basic it is. The capacity of chlorine to kill microorganismsis directly impacted by pH. As it rises, this capacity is heightened too.However, keeping a stable pH level of 7.2 8.0 is recommended to preventcloudy water.
On the other hand, alkalinity determinesthe concentration of alkaline substances, assessing the waters ability towithstand a change in pH. Note that it is the alkalinity that directlydetermines how much acid is required to reduce pH levels, not the pH itself.The suggested range for alkalinity is 80150 parts per million .
Learning how to raise alkalinity in pool without affecting pH is easy when there is a straightforward and savvy approach. Baking soda has a pH level of 8 naturally. Adding it in your pool will increase both pH level and alkalinity, enhancing water clarity, while saving costs.
What Is Pool Ph
Just in case you need a little explanation or reminder on what pH means, lets start with the basics.
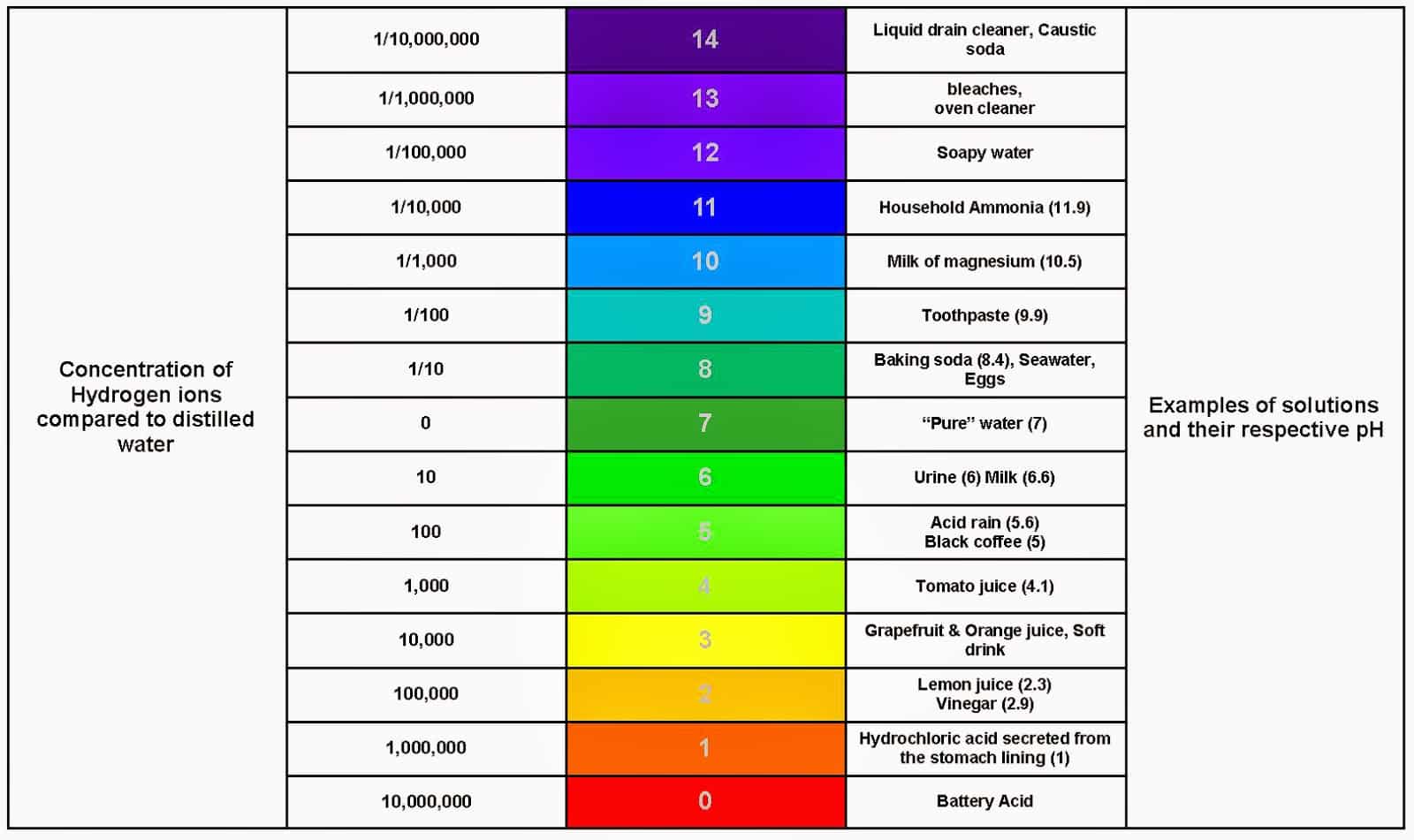
pH is the measure of the acidity or alkalinity of a substance. In this case, the measure of acidity or alkalinity of your swimming pool. Scientifically, it is known as hydrogen ion concentration .
The pH scale runs from 1 to 14. And on thisscale, the most acidic your pool pH can be is 0, while 14 is the most alkalinepH value. However, the recommended pH value for a swimming pool is between 7.2and 7.6. At 7.4, your pool is at the perfect pH.
Also Check: Is Aria Hotel Pet Friendly
Are There Any Risks With Putting Baking Soda In Your Pool
While theres no risk in using baking soda in your pool, there are risks to having high and low alkalinity.
You might notice corrosion in and around your ladders, liner, tiles, or other parts of your pool. This is likely due to low pH and alkalinity for long periods of time.
And if you see scaling or a build-up around your pool equipment, this could be caused by high pH and alkalinity. And it means your water is hard.
All you have to do is keep the alkalinity between 100 and 150 ppm and the pH between 7.4 and 7.6 as often as possible. Test your pool water daily and adjust your chemical levels when needed.
How To Bring Ph Back Down
Balancing your pool can be tricky, and you may find after raising the alkalinity that the pH level is now too high.
You can lower the pH by adding a measured amount of muriatic acid or dry acid to your pool water. Sulfuric acid is another option, but it significantly raises your TDS levels too.
Muriatic acid is highly corrosive so always make sure you take extreme safety precautions when using it. Instructions on the bottle will help you with the correct dosage for your pool, and adding it is simple using a 10:1 solution thats diluted with water.
Dry acid is a milder option that comes in granular or powder form, which makes it slightly easier to work with. Its also more expensive and just as dangerous if mishandled. Once again, its best to follow the instructions on the label.
Be aware that lowering pH will also ultimately lower alkalinity using the methods described above. In that case, you can use baking soda to raise alkalinity with minimal effect on pH.
Finally, as usual, use water testing methods before and after to lower the pH to between 7.4 and 7.6, the ideal range for neutral pool water.
Recommended Reading: Building Your Own Inground Swimming Pool
Are Block Pools Considered Gambling
FACT. Anytime you bet on the outcome of sporting event it is considered gambling. However, CCPD Commander Todd Greenwood says you can avoid legal problems as long as everyone in the pool has an equal opportunity to win, and the person running the pool doesnt take a cut of the pool. Copyright 2021 Scripps Media, Inc.
Why Is My Total Alkalinity Level Too Low
- You may have lowered the alkalinity too much using an alkalinity reducer such as muriatic acid or dry acid. If this is the case, you need to add only a portion of the recommended dosage when lowing your alkalinity, then retest about six hours later.
- You may have backwashed or otherwise drained part or all of the pool water. If you add water to your pool, and the water source has low alkalinity, then this will decrease the overall total alkalinity of your pool. How much the alkalinity decreases depends on how much water you add, how large your pool is, and the water source alkalinity.
- It also may have rained, flooded, or some of the spring snow thaw may have seeped into the pool. Just as a local water source with low alkalinity would lower the total alkalinity of your pool, any other water with low alkalinity will do the same.
Read Also: How To Heat Above Ground Pool
Take Readings Again After 24 Hours
The next day, a full 24 hours since I started my test, the total alkalinity had gone down to 120 ppm. This is not the 100 ppm target value I set, but I was still impressed since 120 ppm is already within the recommended value for a standard swimming pool.
On the other hand, pH was lower, reading 7.1. I didn’t worry too much about this because according to my digital pool water test kit and pool calculator, the lowest it pH can go is 6.8 and still be okay and non-destructive. Plus, the aeration process wasn’t finished.
Since both chemicals were within a reasonable range, all was fine for me. So I waited to take my next reading in the evening, approximately 5 to 6 hours later. At that time, TA was ranging at 106 ppm, and pH had dropped to 6.9.
This means that total alkalinity was inching closer to my target value of 100, despite pH going down.
But Wait What Is Pool Alkalinity
Got your mental lab coat on? You might want to, because this is about to get technical.
Alkalinity is short for total alkalinity, which is a total concentration of dissolved carbonates, bicarbonates, hydroxides, and cyanurates present in the water. You dont have to remember these names, though. Just know that theyre all alkaline substances, meaning that theyre substances whose pH level measures higher than a 7.
In other words, you are not going to find a bag of alkalinity like you would chlorine available at any store . Like pH, it simply doesnt exist.
And while were talking about pH, lets clear up a major misconception.
To keep your circulation system in top shape, youll need a reliable, powerful, and energy-saving pump like the 2 HP Energy Star Variable Speed In Ground Blue Torrent Cyclone Pump to make sure your chemicals are evenly distributed. Plus, it comes with a free warranty, is eligible for rebates, and pays itself off in under a year. According to customer James Robinson, Great price, quick shipping and delivery. Installed it and turned it on, and it was so quiet that we both reached down to feel if it was vibrating! Simple controls. Id buy it again.
You May Like: Concrete Pool Cracks
How To Lower Alkalinity In A Pool
If the alkalinity of the water is too high, i.e. more than 120 ppm, its an easy fix to lower the alkalinity of the water.
First, make sure the water pump is on and take a current pH measurement.
Add one quart of muriatic acid or 2 pounds of dry acid at a time around the perimeter of the pool.
Let the acid mix in the water for about an hour, then test the water again. Maintain a water pH of about 7 and keep adding pool acid in small increments until the correct alkalinity level is reached.
Once the alkalinity level is correct, allow the pH to climb naturally by circulating the water.
Only add soda ash to raise the pH if the pH stops climbing after 1 week.
What Are Alkalinity Enhancers
Alkalinity enhancers are a common chemical available for purchase at pool stores. But if you look at the ingredients of these enhancers, you will notice the main ingredient is sodium bicarbonate, which is more commonly known as drum roll please baking soda!
In reality, using plain old baking soda is a good DIY alkalinity booster. Plus, its cheaper and has the same effect as store-bought alkalinity enhancers.
Also Check: Will Pool Water Kill Lice
What Causes Low Alkalinity In Pool
Its very normal to experience a drop in alkalinity levels in your pool over the season. For the most part, this is due to natural causes such as evaporation or agitation of your water. Throughout the hot summer season, your pool will release dissolved carbon dioxide in a process known as water degassing. This directly impacts total alkalinity, causing it to fall below 80 ppm if left untreated.
Other causes of low alkalinity include:
- overuse of alkalinity lowering products
- using too many chlorine tablets with low pH
- excessive rainwater diluting the pool
- draining or backwashing pool water
What Is The Ideal Alkalinity In Pool

The ideal alkalinity range of a balanced pool should be between 100 and 140 parts per million . Its perfect for keeping it between this range as it ensures appropriate buffer and positive chlorine effects.
Sodium and Calcium Hypo-Chlorite pool shocking products perform well in the lower end of this alkalinity range, while chlorine gas, dichlor and trichlor perform better in the higher end.
Anything below 80 ppm or above 150 ppm for most swimming pool require immediate balancing. These alkalinity levels potentially affect the pools LSI reading significantly and put your pool surfaces and equipment at risk of degradation.
A standard alkalinity test gives you the pools total alkalinity, and that is everything you need. But apart from knowing the TA, you might want to tell the carbonate alkalinity level.
You get this by subtracting CA from TA to get them corrected or true TA, the alkalinity level you need to adjust.
You May Like: Can Lice Live In Swimming Pools
Set A Target Value For Total Alkalinity
In the same example above, I set my target value of TA at 100 ppm. It is important to note that both the levels of TA and pH reduce when acid is added. A target value of 80 ppm or 90 ppm should be just fine, but I anticipated that the levels of pH would drop way below 7.0 when I add a strong acid, so my experience told me that 100 ppm was ideal for raising the level of pH later on through aeration.
Test The Alkalinity Of Your Pool
After adding the product and waiting about 6 hours for it to disperse and dissolve in your swimming pool, its time to start testing. Make sure you have a reliable testing kit for both total alkalinity and pH.
You can use pool water testing strips or a liquid water testing kit.
man testing for low alkalinity in swimming pool water
Begin to take readings after 6 hours, then 24 hours, then 48 hours to gauge if there are any small changes over this period and to ensure the chemicals are balanced. Make sure that both the total alkalinity and pH values are within optimal ranges. If so, your job is done!
Also Check: Mandalay Bay Pool Times
How Much Baking Soda
One of the potential disadvantages of using baking soda instead of a commercial alkalinity increaser is that baking soda does not come with instructions for use on pools. However, that should not keep you from using it to increase your alkalinity levels.
To calculate the amount of baking soda you want to use, you should first determine how many gallons of water are in your pool. Then, add 1.5 pounds of baking soda for every 10,000 gallons of water. This will increase the pools total alkalinity by 10 ppm.
For example, if your pool houses 20,000 gallons of water and your test kit has indicated an alkalinity of 50 ppm, you need to raise your alkalinity by at least 30 ppm. With a pool of 20,000 gallons of water, every three pounds of baking soda will raise the water roughly 10 ppm. Therefore, you need to add about nine pounds of baking soda to a 20,000-gallon pool to increase the alkalinity by 30 ppm. This will put the water in the acceptable alkalinity range of 80 -120ppm.
Once youve calculated how much to use, add the baking soda directly into the water. Let it dissolve and allow enough time for it to fully distribute throughout the pool. This will usually take a minimum of twenty minimums with your pump on. Use your test kit to check the levels in the water and add more baking soda as needed.
Pool Ph And Alkalinity Issues
It is not uncommon to have unusual variations in relation to the pH or total alkalinity in your swimming pool. With such a great diversity in water types from region to region, and even within individual communities, achieving a proper balance can be challenging. While its always better to have pH and alkalinity levels on the high end than the low end, there are steps you can take to achieve a better balance.
Here are some of the most common issues with pool pH and alkalinity and how to fix them:
You May Like: Build An Inground Pool
Keep An Eye On Alkalinity
Are your pH levels still too low after adding in the soda ash? Try testing the alkalinity of your water!
If the pool waters alkalinity is too low, the pH of your pool water will fluctuate. Your waters alkalinity should be between 80 ppm and 150 ppm!
Once youve discovered that your pools alkalinity is too low, add in 1.4 pounds of baking soda for every 10,000 gallons of water in your pool.
Let the baking soda circulate through your pool with the pump turned on for around one hour before you test the alkalinity again. Not only does baking soda increase the alkalinity of your pool water, but it also raises the pH levels!
However, be aware that youre not adding too much baking soda into your pools water. If youre adding in baking soda after youve added in soda ash, know that soda ash also increases the alkalinity in your pool. Its much harder to lower your alkalinity levels than it is to raise them!
Dilute Muriatic Acid As Directed And Add It To The Water
The pool in my example holds 20,000 gallons. To get my target value of 100 ppm, I needed to add about 2 quarts of muriatic acid.
I usually distribute the muriatic acid solution slowly around the pool while the pump is running high to increase the effect.
Some pool managers prefer adding the muriatic acid solution in one spot. Personally, I don’t like to do this because acid can damage pool parts when it is concentrated in one region.
The natural process of aeration begins after you add the muriatic acid and the levels of pH and TA have fallen and settled. During the aeration process, the levels of pH alone will rise gradually until it reaches the recommended level.
Also Check: Pentair Heater/chiller Combo
The Roles Of Ph In Water Chemistry
Chlorine strength
Managing pH is a critical component of maintaining healthy and balanced water chemistry. It is so important because pH is a driving factor in the Langelier Saturation Index , and also because pH determines how effective chlorine will be in non-stabilized pools. So in pools with no cyanuric acid , the lower the pH, the stronger chlorine will be, because pH controls the dissociation equilibria between strong and weak chlorine:
HOCl H+ + OCl-Hypochlorous Acid dissociates into Hydrogen and Hypochlorite Ion
Again, pH controls the strength of chlorine because the lower the pH, the higher the concentration of Hydrogen. That means a higher % of strong HOCl vs. weak OCl-. This is not as relevant in pools with CYA, because isocyanurates fundamentally change pool chemistry.
Calcium carbonate solubility
While the LSI determines when water is oversaturated with calcium carbonate , pH is a major factor on the LSI. And since LSI violations occur locally, not universally throughout a pool, pH matters a lot. For instance, there’s a chart below that shows the equilibria of alkalinity types. When the pH gets to 8.2 or 8.3, it becomes very difficult for calcium to stay in solution, because carbonate alkalinity binds really well with calcium . So when your pH gets too high, calcium tends to precipitate out of solution even if the overall LSI of your pool is balanced. We see this phenomenon cloud up pools, like when someone adds soda ash too quickly.
CO2 determines pH
